Introduction
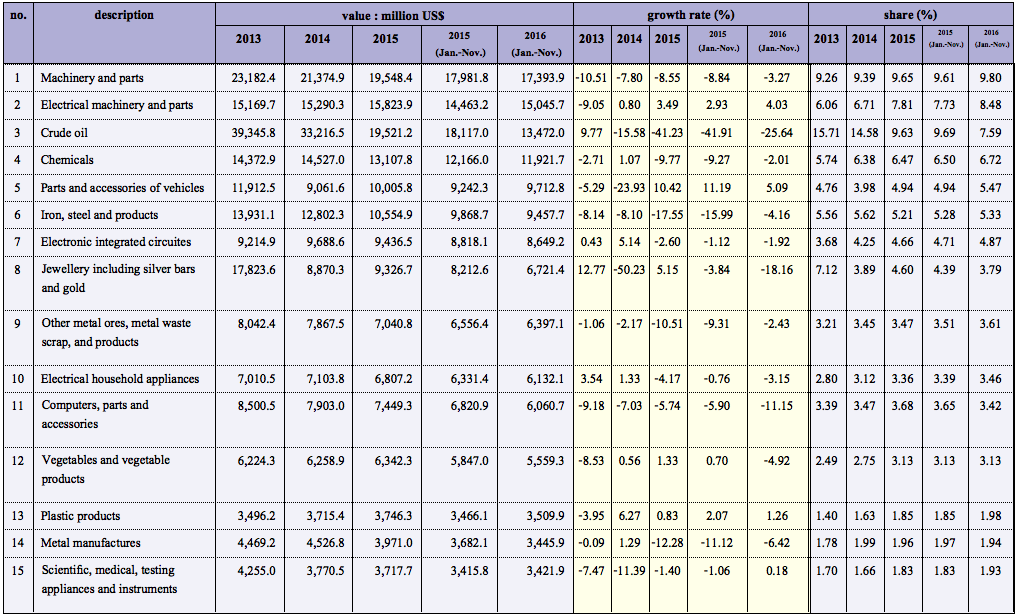
Until June 9, 2022, cannabis and hemp had been classified as a category 5 narcotic under Thailand’s 1979 Narcotics Act. For many years, all activities related to the plants and their derivatives was heavily restricted until regulatory liberalization began in February 2019 with an amendment to the Narcotics Act. Over the next few years, restrictions on specific types or uses of cannabis and hemp were gradually loosened, and then Thailand became the first country in Asia to permit growing, selling, and using the plant for both medical and other permitted purposes when it delisted both cannabis and hemp plants and their unprocessed parts from the Narcotics Code on June 9, 2022.
This delisting now allows the private sector to grow, possess, sell, and use locally cultivated cannabis and hemp plants without a license. However, the Narcotics Code still regulates cannabis and hemp extracts containing tetrahydrocannabinol (THC) of more than 0.2%. Making or handling a substance with THC over this limit still requires a license, with limited exceptions.
However, the breadth of this liberalization will likely be tempered in the future. The draft Cannabis and Hemp Act, an omnibus law currently under the Thai Parliament’s consideration, is expected in the months to come. This act will specifically govern cannabis and hemp, including by imposing controls such as new restrictions or an enhanced licensing regime.
Most of the product classifications discussed here are based upon THC content, and on the presence of cannabidiol (CBD) in the products. As THC is considered a psychoactive substance—in comparison to CBD, which is not psychotropic and has been applied in various therapeutic indications—a higher THC content may therefore be associated with a greater risk of abuse, and thus stricter regulations will likely apply in Thailand. Under the most recent legal amendments, Thai law defines cannabis as in the Cannabis sativa L. subsp. indica, and hemp as Cannabis sativa L. subsp. sativa with no more than 1.0 % THC by dry weight in its leaves and inflorescence.
The key regulatory body for cannabis and hemp-related products is the Thai Food and Drug Administration (Thai FDA), a government agency operating under supervision of the Ministry of Public Health. Working closely with the Narcotics Control Committee, the Thai FDA is mainly responsible for granting and administering licenses and post-marketing control, among others. Representatives of the Thai FDA also sit on most of the national policymaking committees. One of the other Thai government bodies involved in the regulation and control of cannabis and hemp is the Department of Agriculture (under the Ministry of Agriculture and Cooperatives), which administers the rules on the importation of plant seeds (e.g., cannabis and hemp seeds).
Product Classification
Activity-Focused Regulatory Overviews
Cultivating Cannabis/Hemp
Following their June 9, 2022, delisting, Thai nationals may freely cultivate cannabis and hemp plants and trade unprocessed plant parts and crude resins in Thailand without a license.
As stated earlier, however, the current breadth of this liberalization is expected to be temporary. Upon promulgation of the current draft Cannabis and Hemp Act, the industry is likely to face new restrictions and an enhanced licensing regime.
Companies and importation activities are subject to various regulations. For companies, agricultural activities (including growing cannabis/hemp) are still reserved for majority Thai-owned companies.
Importation of cannabis/hemp plants and most unprocessed parts is still prohibited, except for cannabis/hemp seeds, which may be imported under the rules and procedures of the Plant Act (1975) and Plant Quarantine Act (1964).
Producing or Importing Cannabis/Hemp Extracts and Seed Oils
Industrial extraction of cannabis or hemp still requires a Thai FDA license under the Narcotics Code.
After the June 9, 2022, delisting, the following products are now deregulated:
- Extracts containing 0.2% or less THC by weight produced from domestically grown plants and under license from the Thai FDA, and
- Seed oils produced from domestically grown seeds.
These deregulated products may be sold, possessed and consumed without a license under the Narcotics Code. Howver, handling any extract or oil that does not fall within the conditions set out above requires a license from the Thai FDA under the Narcotics Code.
Importation. Importation of any extract or oil is still prohibited.
Producing or Importing Medical Cannabis/Hemp and Other Herbal Products
Production. Irrespective of THC content, medical cannabis/hemp (including Thai traditional medicines and cannabis oils) and other herbal products (including herbal cosmeceuticals and nutraceuticals) now fall under the purview of the Herbal Product Act (2019). The act, along with its subsequent implementing regulations, provides licensing and product registration requirements for domestic production and distribution of these products.
Importation. Importation of these products is still largely prohibited with a few exceptions: Thai government agencies importing goods for treatment of patients, public academic institutions importing for research purposes, and anyone importing for human clinical trials, subject to the licensing procedures of the Thai FDA under the Herbal Products Act.
Producing or Importing Modern Drugs Formulated with Cannabinoids
Modern drugs formulated with cannabis, hemp extract, or other cannabinoids require regulatory review and licensing from the Thai FDA under the Drug Act. The licensing rules and procedures are already in place through existing ministerial regulations and notifications. Notably, a drug manufacturer or importer license is required for the business operator, and a drug registration certificate is required to market a particular modern drug formulation.
Producing or Importing Cosmetics with Hemp- and Cannabis-based Ingredients
Production. Domestically produced cosmetics may contain any or combination of hemp seed oils, hemp seed extracts, parts of hemp (except inflorescence), parts of cannabis (except inflorescence and seeds), and CBD extracts, provided that the raw materials are obtained from plants grown in Thailand. The production requires a Thai FDA license under the Cosmetics Act. Additionally, the products themselves must comply with the specifications prescribed by the relevant ministerial notifications.
Cannabis seeds, cannabis seed oils, and inflorescences of cannabis and hemp, regardless of their origins, are prohibited from cosmetic product formulations.
Importation. Importation of cosmetic products containing hemp- or cannabis-based ingredients is still prohibited.
Producing or Importing Food, Beverages, Food Supplements, or Food Additives with Hemp- and Cannabis-based Ingredients
Production. Food products may contain hemp seeds, hemp seed oils, hemp seed proteins, parts of hemp (except inflorescence), parts of cannabis (except inflorescence and seeds), and CBD extracts, provided that these are obtained from plants grown in Thailand. For domestically produced goods, a Thai FDA license is required under the Food Act, and the product must comply with the relevant ministerial notifications.
While this allowance is applicable to many food products, there are exceptions. For example, no part of cannabis or hemp may be added to food for infants, food for young children, or energy drinks. In addition, certain foods do not allow addition of cannabis and hemp. Certain other foods allow use with conditions. Because of this variation in rules for different kinds of food, it is important to check the relevant regulations to ensure compliance.
Cannabis seeds, cannabis seed oils, and inflorescences of cannabis and hemp are not allowed as ingredients in any food product.
Importation. Importation of food products containing hemp- or cannabis-based ingredients is still prohibited—even if the cannabis- or hemp-based ingredients came from plants grown in Thailand.
Ready-to-Eat Foods and Beverages
Thai restaurants and cafés are allowed to use cannabis parts (except inflorescence and seeds) and hemp parts (except inflorescence) in their recipes.
Crop and processing infrastructure
The Cannabis and Hemp Act drafting committee is planning to organize cultivation into three classes. Small cultivators, with no more than 10 plants per household, may not have to pay fees but will have to notify the Thai FDA of their growing activities. Medium cultivators, with less than 32,000 square meters (<20 rai) under cultivation, will have to go through additional bureaucratic procedures and pay additional fees. Large cultivators, with over 32,000 square meters (>20 Rai) under cultivation, will be considered commercial cultivators and require FDA approval. Fees for large cultivators are likely to include both monthly and annual payments.
The committee has decided that modern medical practice and traditional physicians, and other healthcare professionals cultivating cannabis for medical purposes, will only be required to notify the FDA of their activities (similar to the requirements for small cultivators).
The Narcotics Division of the Thai FDA published handbooks on growing and handling cannabis crops in 2021. Cultivators can choose among outdoor, semi-outdoor, and indoor cultivation. Outdoor cultivation is described as being low-cost and suitable for Thai cannabis varieties, with the drawbacks of limited pest-control options and harvesting taking place annually. On the other hand, indoor cultivation comes with higher costs but with the advantages of better quality control and year-round harvesting. As for the import of cultivation and processing infrastructure, such as equipment for crop maintenance, testing, extraction, formulation, and packaging, relevant regulations are in place and need to be considered.
Intellectual Property
Patents
On January 28, 2019, the Thai government issued an ordinance to reject a number of Thai patent applications filed by foreign applicants, all of which related to medical formulation or use of cannabis-derived active ingredients. The ordinance was issued to annul any private exclusive rights that could block access to medical cannabis following its legalization, and it was terminated on February 19, 2019, when cannabis legalization took effect. Since then, patentability has once again been examined against the previously established rules of the Patents Act. This means that generally, an application related to cannabis is eligible for a patent if it is not for recreational purposes; not a diagnostic or therapeutic method; and not the plant itself, its part, or crude extract.
Trademarks
Generally, trademark protection for goods or services related to legalized use of cannabis or hemp is available in Thailand, as long as the trademark meets the usual requirements for registration.
Key Dates
| April 28, 1979 | The Narcotics Act (No. 1) took effect, declaring cannabis and hemp category 5 narcotics. |
| January 1, 2018 | The Hemp Regulation RE: Application of the License and Issuing of Licenses to Manufacture, Disposal or Possession of Hemp came into effect, allowing cultivation and processing of hemp only for industrial such as textiles, paper industries, etc. or non-commercial uses, such as consumption by indigenous groups or research and development. Nonetheless during 2018 until January 29, 2021, only state agencies were able to obtain the licenses in relation to hemp. |
| Janurary 28, 2019 | The National Council for Peace and Order’s Ordinance No. 1/2562 became effective, rejecting Thai patent applications related to cannabis (see also Intellectual Property, above). |
| February 19, 2019 | The Narcotics Act amendment (No. 7) came into effect, legalizing medical use of substances listed as category 5 narcotics (e.g., cannabis, hemp, and kratom). |
| March 30, 2019 | A ministerial notification took effect setting out 16 cannabis-containing Thai traditional medicine formulations that may be consumed for therapeutic purposes without having to be registered with the Thai FDA, as long as manufacturers obtain a narcotic production license from the Thai FDA [overridden by the Key Event of June 9, 2022, below]. |
| May 21, 2019 | End of the “amnesty period,” during which persons possessing cannabis for certain purposes (medical, research, etc.) could declare their possession to the Thai FDA and be exempted from criminal punishment. |
| August 7, 2019 | The Government Pharmaceutical Organization launched the first batch of medical CBD oil, THC oil, and CBD: THC oil (4,500 units of 5 milliliters each) for the special access scheme (SAS) in public hospitals. |
| February 15, 2020 | The Minister of Public Health updated the list of cannabis-containing Thai traditional medicine formulations that may be consumed for therapeutic purposes and/or research purposes, without having to be registered with the Thai FDA [overridden by the Key Event of June 9, 2022, below]. |
| December 14, 2020 | The “delisting” ministerial notification became effective, carving out domestically produced items containing certain cannabis and hemp plant parts, and CBD extract with less than 0.2% THC by weight, from the scope of Narcotics Act (see also Product Classification, above). Importation of these products still requires an importation license as prescribed in the Narcotics Act [overridden by the Key Event of June 9, 2022, below]. |
| January 29, 2021 | The Hemp Regulation RE: Application of the License and Issuing of Licenses to Manufacture, Import, Export Disposal or Possession of Hemp took effect, allowing cultivation for production of modern drugs, herbal products, cosmetics, and food products, as well as for household and certain other uses. This allowed hemp to be used in healthcare- related products. Private entities established in Thailand and groups of farmers (e.g., cooperatives or community enterprises) can obtain licenses without forming a partnership with a state agency. |
| June 4, 2021 | Eight herbal formulas containing cannabis as an active ingredient are listed in the National List of Essential Drugs, which identifies both modern drugs and herbal products deemed necessary for the prevention and treatment of major health problems in Thailand. It also provides a drug reimbursement mechanism for government hospitals. |
| August 11, 2021 | Cannabis and hemp seeds become controlled seeds under the Plants Act (1975). The seeds must exhibit at least 70 percent germination and 99 percent purity. Licensed traders (i.e., importers, collectors) must follow the requirements prescribed in the Plants Act and its bylaws. |
| August 24, 2021 | Kratom is removed from the list of category 5 narcotics, (some parts of cannabis and hemp plants and their derivative products remain on the list—see December 14, 2020). |
| October 25, 2021 | Cannabis L. genus is recognized as eligible for protection as a new plant variety under the Plant Variety Protection Act (1999). |
| November 26, 2021 | The Cannabis Regulation RE: Application of the License and Issuing of Licenses to Manufacture, Import, Export, Dispose or Possession of Cannabis takes effect. |
| December 9, 2021 | The Narcotics Code becomes effective, replacing and consolidating the previous Narcotics Act and Psychotropic Substances Act, among others. The Narcotics Code has transitional provisions that maintain the effects of regulations and validity of licenses previously issued under the Narcotics Act. |
| June 9, 2022 | Cannabis and hemp plants are delisted from the Narcotics Code. Thai nationals may grow the plant and handle unprocessed parts without a license. However, extracts and seed oils remain governed by the Narcotics Code. Finished products may contain cannabis or hemp under the regulation of their respective laws. |
| June 16, 2022 | The Minister of Public Health issues an official notification under the Act on Protection and Promotion of the Knowledge on Thai Traditional Medicines (1999). The notification prohibits selling cannabis to minors (under the age of 20), use of cannabis by pregnant and breastfeeding women, and smoking cannabis in public. |
| Forthcoming | The Cannabis and Hemp Act will likely institute new restrictions and an enhanced licensing regime. |
© 2021 Tilleke & Gibbins International Ltd.